Taxonomic group: plant / Streptophyta
(Phylum: Streptophyta)
Organ / tissue: leaf
The structure was elucidated in this paperNCBI PubMed ID: 10489886Publication DOI: 10.1007/BF02979070Journal NLM ID: 8000036Publisher: Pharmaceutical Society of Korea
Correspondence: Whang WK <Whang-wk

cau.ac.kr>
Institutions: College of Pharmacy, Chung-Ang University, 156-756, Seoul, South Korea
A study was carried out to evaluate flavonol glycosides in leaves of Symplocarpus renifolius (Araceae). From the water fraction of the MeOH extract, three new flavonol glycosides were isolated along with three known compounds, kaempferol-3-O-β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl-7-O- β-D-glucopyranoside, quercetin-3-O-β-D-glucopyranosy-l-(1→2)-β-D-glucopyranoside, and caffeic acid. The structures of the new flavonol glycosides were elucidated by chemical and spectral analyses as quercetin-3-O-β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl-7-O-β-D-glucopyranoside, isorhamnetin-3-O-β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl-7-O-β-D-glucopyranoside, and quercetin-3-O-β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl-7-O-(6″″-trans-caffeoyl)-β-D-glucopyranoside.
quercetin, kaempferol, leaves, flavonol glycosides, Araceae, Symplocarpus renifolius, isorhamnetin derivatives
Structure type: monomer ; 463 [M-H]-
Location inside paper: compound III-P, table 1(III-P), p. 427 (compound III-P)
Compound class: flavonol glycoside
Contained glycoepitopes: IEDB_142488,IEDB_146664,IEDB_983931,SB_192
Methods: 13C NMR, 1H NMR, EI-MS, IR, FAB-MS, TLC, enzymatic hydrolysis, HPLC, alkaline hydrolysis, UV, optical rotation measurement, HCl hydrolysis, sulfuric acid hydrolysis
Synthetic data: chemical
Comments, role: NMR temperature was not specified; product of partial acid hydrolysis of ID 62906
Related record ID(s): 62904, 62905, 62906, 62908, 62909
NCBI Taxonomy refs (TaxIDs): 477955
Show glycosyltransferases
NMR conditions: in DMSO-d6
[as TSV]
13C NMR data:
Linkage Residue C1 C2 C3 C4 C5 C6 C7 C8 C9 C10 C11 C12 C13 C14 C15 C16
7 bDGlcp 100.3 73.2 77.2 69.9 76.5 60.9
xXQuercetin - 147.9 135.9 175.9 160.3 98.9 162.8 94.5 155.7 104.7 121.9 115.5 145.0 147.8 115.5 120.1
1H NMR data: present in publication
|
13C NMR data:
| Linkage | Residue | C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | C9 | C10 | C11 | C12 | C13 | C14 | C15 | C16 |
|---|
| 7 | bDGlcp | 100.3 | 73.2 | 77.2 | 69.9 | 76.5 | 60.9 | |
| | xXQuercetin |
| 147.9 | 135.9 | 175.9 | 160.3 | 98.9 | 162.8 | 94.5 | 155.7 | 104.7 | 121.9 | 115.5 | 145.0 | 147.8 | 115.5 | 120.1 |
|
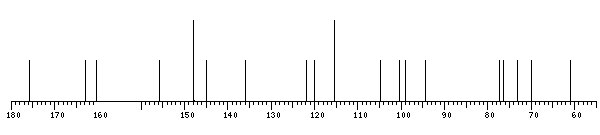
The spectrum also has 1 signal at unknown position (not plotted). |
There is only one chemically distinct structure:
 report error
report error report error
report error